PLASMA DONATION WAVE 1
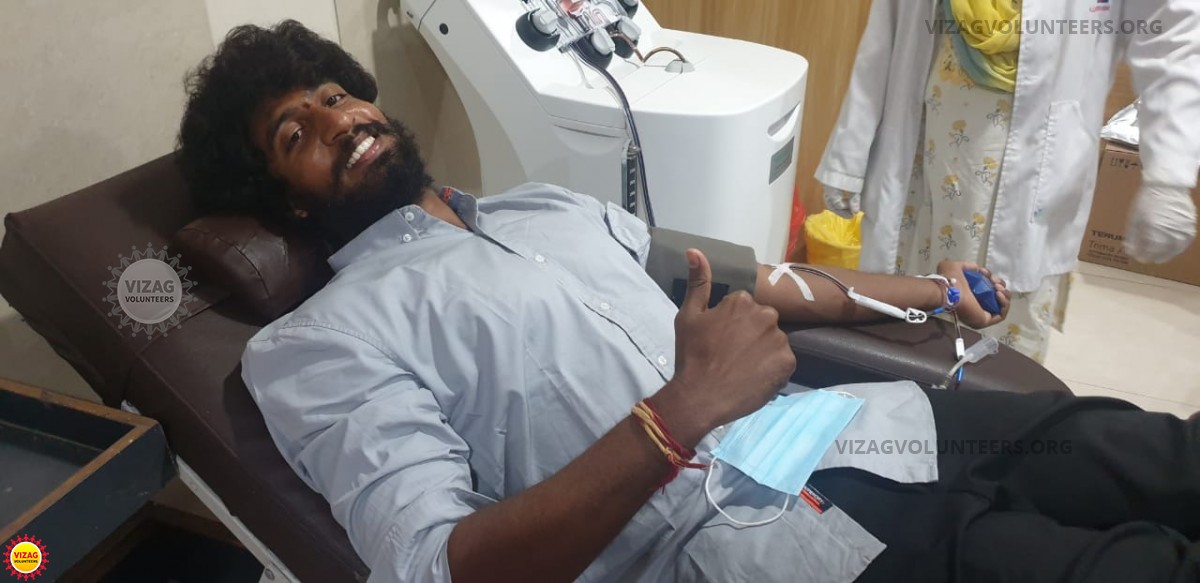
The covid-19 treatment guidelines considered plasma therapy to be one of the effective lines of treatment when a person was in a severe condition of Covid-19. So we started our research on plasma therapy...
Read MoreThe covid-19 treatment guidelines considered plasma therapy to be one of the effective lines of treatment when a person was in a severe condition of Covid-19. So we started our research on plasma therapy and thought to bring it to Vizag as soon as possible as there were many deaths with every passing day. We also thought to bring plasma therapy to everyone in need without differences like rich or poor. We were inspired by the Delhi model of operations for creating a plasma bank. Whoever wanted to donate plasma could donate voluntarily, just like blood donations, for every request raised, a quick disbursement of plasma unit for the recipient was done.
Our main motives were To identify a blood bank and convert it as a plasma bank to respond quickly to every request raised. To curtail the rising black market. Never connect donor or recipient. To motivate people on voluntarily plasma donations.
The covid-19 treatment guidelines considered plasma therapy to be one of the effective lines of treatment when a person was in a severe condition of Covid-19. So we started our research on plasma therapy...
Read More
Our country has met with the second wave of catastrophe no one has imagined, and Vizag has also faced this COVID wave. Many people got affected and started admitting to hospitals. People began requesting...
Read MoreDesigned By F9tech. All Rights Reserved.